Experimental Pharmacotherapy: Dietrich Lab "Identification of Pharmacological Targets in Pathophysiological Mechanisms"
Experimental Pharmacotherapy: "Identification of Pharmacological Targets in Pathophysiological Mechanisms"
Head: Prof. Dr. Alexander Dietrich deutsche Version
MD/PhDStudents: Philipp Alt (pharmacist), Fabienne Geiger (M. Sc. in Biochemistry), Isabel Müller (M. Sc. in Chemistry), Suhasini Rajan (M. Sc. Translational Medical Research), Lena Schaller (M. Sc. in Medical Science)
Technical Assistants: Bettina Braun
In our research group for Experimental Pharmacotherapy we focus on the identification of pharmacological targets in pathophysiological mechanisms. In the future, we would like to activate or inhibit these targets in animal models and in patients. We are using gene-deficient or knock-out mouse models with deleted genes of interest to understand physiological and pathophysiological functions of different target proteins in the organism.
Next to receptors ion channels are important drug targets. Therefore, we analyze functions of „transient receptor potential“ (TRP) channels (e.g. classical TRP 6 (TRPC6) Fig. 1) for the development of diseases in lung (1) and other tissues.
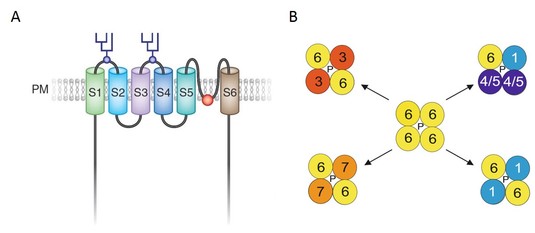
Fig 1: (A) Structural features and topology of the TRPC6 channel in the plasma membrane (PM). Transmembrane regions (S1 through S6) and the conserved predicted pore domain between S5 and S6 are indicated. Two glycosylated sites in TRPC6 are marked by covalently bound carbohydrates (from 2).
(B) Functional TRPC channels form tetramers composed of four identical or different TRPC channel monomers (1, 3, 4, 5, 6, 7). Through a functional pore (P) of these channel tetramers unselective cations e.g. Na+ and Ca2+ enter the cell (from 3). The molecular activation mechanism of TRPC6 channels is analyzed by Förster resonance energy transfer (FRET) in the lab (4).
As a member of the German Lung Center (DZL) we were able to detect numerous roles for TRP channels in the lung. In primary lung fibroblasts TRPC6 is up-regulated after application of the cytokine “transforming growth factor beta (TGF-β)” and supports myofibroblast differentiation and the development of lung fibrosis (5). Store-operated Ca2+ influx in these cells was, however, not dependent on TRPC channels (6). Moreover, we were able to identify an essential role of TRPC6 for acute hypoxic pulmonary vasoconstriction (aHPV) (7). Most interestingly, TRPC6-deficient mice lacking aHPV showed severe arterial hypoxemia after blockage of parts of the lung. In the future, TRPC6 activators may present important therapeutic agents for patients with lung sepsis, pneumonia or systemic inflammation to avoid arterial hypoxemia (Fig. 2).
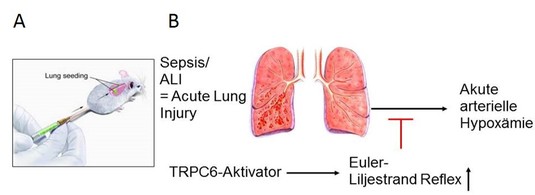
Fig. 2: (A) Injection of drugs in living mice and accumulation in the lung.
(B) Possible future therapeutic intervention by TRPC6 activators to activate acute HPV during partial blockage of the lung and to avoid acute arterial hypoxemia.
Next to smooth muscle cells, endothelial cells are essential for pulmonary gas exchange. We were able to show recently that TRPC6-deficient lungs are protected from ischemia-reperfusion injury (IRI). Lung IRI might occur if lungs are not perfused during transportation. We demonstrate that TRPC6-induced decreases in lung endothelial permeability are essential for IRI and are suppressed by TRPC6 deficency (8). Most interestingly, a specific blockage of TRPC6 by an inhibitor provided by Prof. Michael Schaefer’s lab, University of Leipzig, was able to reduce IRI (9) and might protect isolated lungs intended for transplatation in the future.
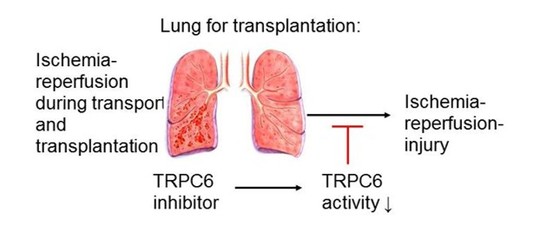
Fig 3: TRPC6-inhibitors applied to isolated lungs intended for transplantation might help to reduce lung ischemia-reperfusion injury.
Another TRP channel of the vanilloid family (TRPV4) is involved in the production of surfactant protein C (SP-C) in alveolar type II (ATII) cells of the lung epithelium for protection and increased oxygen uptake. After ablation of TRPV4 channels in these cells, aged mice develop emphysema-like changes, which - in clear contrast to TRPC6 - increase ischemia-reperfusion-induced edema formation. Moreover, production of a water permeable channel (aquaporin 5 (AQP-5)), barrier function as well as migration was reduced in alveolar type I (ATI) cells (10).
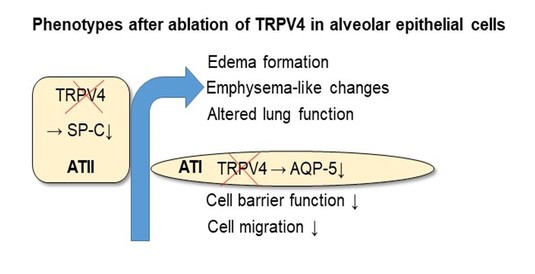
Fig. 4: Ablation of TRPV4 channels in lung epithelium reduced production of surfactant protein C (SP-C) in alveolar type II (ATII) cells, inducing emphysema-like changes and altered lung function in aged mice. Moreover, a water permeable channel aquaporin 5 (AQP-5) was reduced and cell barrier function and migration was inhibited in alveolar type I (ATI) cells. In clear contrast to TRPC6-deficient lungs, TRPV4-deficient lungs show higher ischemia-reperfusion-induced edema formation (10 and Video).
Other TRP channels like TRPA1 (11), TRPM2 and TRPV4 are also expressed in different lung tissues. Their physiological roles are explored in the Transregional Collaborative Research Centre 152 TRiPs to Homeostasis. Another possible function of TRP channels as sensors for toxicant (12) and radiation (13) in the lung will be elucidated in the just established Research Training Group for deciphering therapeutic targets in lung toxicology.
TRP channels are important signaling molecules for kidney function (2, 14), in allergic responses of the lung airways (15), skeletal muscle function (16), for intestinal motility (17), light-touch sensation (18), neuronal transmission (19) and behavior (20). Most interestingly, “loss of function” mutations in TRPC6 also support the development of autism (21). Work on all these topics are executed in close collaboration with research groups around the world. We use molecular biology and cell biology techniques in combination with intracellular biosensors to compare gene-deficient cells from different mouse models.
Selected references:
1. Dietrich, A. (2019) Modulators of Transient Receptor Potential (TRP) Channels as Therapeutic Options in Lung Disease. Pharmaceuticals 12:23. doi: 10.3390/ph12010023.
2. Dietrich, A., Chubanov, V., Gudermann, T. (2010). Renal TRPathies, J. of Am. Soc. Nephrol. 21: 736-744.
3. Dietrich, A, Gudermann, T. (2014) TRPC6. Handbook of Experimental Pharmacology 222, Springer Verlag Heidelberg, 157-188.
4. Fiedler, S., Storch, U., Erdogmus, S., Gudermann, T., Mederos Y Schnitzler, M., Dietrich, A. (2019). Small Fluorescein Arsenical Hairpin-Based Förster Resonance Energy Transfer Analysis Reveals Changes in Amino- to Carboxyl-Terminal Interactions upon OAG Activation of Classical Transient Receptor Potential 6. Mol Pharm 96(1):90-98. doi: 10.1124/mol.119.115949.
5. Hofmann, K., Fiedler, S., Vierkotten, S., Weber, J., Klee, S., Jia, J., Zwickenpflug, W., Flockerzi, V., Storch, U., Yildirim, A.Ö., Gudermann, T., Königshoff, M., Dietrich, A. (2017). Classical transient receptor potential (TRPC6) channels support myofibroblast differentiation and development of experimental pulmonary fibrosis. Biochem Biophys Acta 1863: 560-568.
6. Bendiks, L., Geiger, F., Gudermann, T., Feske, S., Dietrich, A. (2020). Store-operated Ca2+ entry in primary murine lung fibroblasts is independent of classical transient receptor potential (TRPC) channels and contributes to cell migration. Sci Rep. 2020 Apr 22;10(1):6812
7. Weissmann*, N., Dietrich*, A., Fuchs, B., Kalwa, H., Ay, M., Dumitrascu, R., Olschewski, A., Mederos y Schnitzler, M., Ghofrani, H.A., Schermuly, R.T., Pinkenburg, O., Seeger, W., Grimminger, F. and Gudermann, T. (2006). Classical transient receptor potential channel 6 (TRPC6) is essential for hypoxic pulmonary vasoconstriction and alveolar gas exchange. Proc. Natl. Acad. Sci. U.S.A. 103, 19093-19098. *equal contributing and corresponding authors.
8. Weissmann, N., Sydykov, A., Kalwa, H., Storch, U., Fuchs, B., Mederos y Schnitzler, M., Brandes, R.P., Grimminger, F., Meissner, M., Freichel, M., Offermanns, S., Veit, F., Pak, O., Krause, K.-H., Schermuly, R.T., Brewer, A.C., Schmidt, H.H.H.W., Seeger, W., Shah, A.M., Gudermann, T., Ghofrani, H.A. & Dietrich, A. (2012). Activation of TRPC6 channels is essential for ischemia–reperfusion-induced lung edema in mice. Nature Commun. 3: 649.
http://www.nature.com/ncomms/journal/v3/n1/full/ncomms1660.html
9. Häfner, S., Burg, F., Kannler, M., Urban, N., Mayer, P., Dietrich, A., Trauner, D., Broichhagen, J., Schaefer. M. (2018). A (+)-Larixol Congener with High Affinity and Subtype Selectivity toward TRPC6. ChemMedChem. 13:1028-1035. doi: 10.1002/cmdc.201800021.
10.Weber, J., Rajan, S., Schremmer, C., Chao, Y.K., Krasteva-Christ, G., Kannler, M., Yildirim, A.Ö., Brosien, M., Schredelseker, J., Weissmann, N., Grimm, C., Gudermann, T., Dietrich, A. (2020). TRPV4 channels are essential for alveolar epithelial barrier function as protection from lung edema. JCI Insight 5:e134464. doi: 10.1172/jci.insight.134464.
11. Kannler, M., Lüling, R., Yildirim, A.Ö., Gudermann, T., Steinritz, D., Dietrich, A. (2018). TRPA1 channels: expression in non-neuronal murine lung tissues and dispensability for hyperoxia-induced alveolar epithelial hyperplasia. Pflügers Arch. 470: 1231-1241.
12. Dietrich, A., Steinritz, D., Gudermann, T. (2017). Transient receptor potential (TRP) channels as molecular targets in lung toxicology and associated diseases. Cell Calcium 67:123-137. doi: 10.1016/j.ceca.2017.04.005.
13. Käsmann, L., Dietrich, A., Staab-Weijnitz, C.A., Manapov, F., Behr, J., Rimner, A., Jeremic, B., Senan, S,, De Ruysscher, D., Lauber, K., Belka. C. (2020). Radiation-induced lung toxicity - cellular and molecular mechanisms of pathogenesis, management, and literature review. Radiat Oncol 15: 214. doi: 10.1186/s13014-020-01654-9.
14. Kalwa, M., Storch, U., Demleitner, J., Fiedler, S., Mayer, T., Kannler, M., Fahlbusch, M., Barth, H., Smrcka, A., Hildebrandt, F., Gudermann, T. and Dietrich, A., (2015). Phospholipase C epsilon-induced TRPC6 activation: A common but redundant mechanism in primary podocytes. J Cell Physiol 230:1389-1399.
15. Sel, S., Rost, B., Yildirim, A.Ö., Sel, B., Kalwa, H., Fehrenbach, H., Renz, H., Gudermann, T., Dietrich, A. (2008). Loss of classical transient receptor potential 6 reduces allergic airway response. Clin Exp Allergy 38, 1548-1558.
16. Zanou, N., Shapovalov, G., Louis, M., Tajeddine,N., Gallo, C., Van Schoor, M., Anguish, I., Cao, M.L., Schakman, O., Dietrich, A., Lebacq, J., Ruegg, U., Roulet, E., Birnbaumer, L., Gailly, P. (2010). Role of TRPC1 channels in skeletal muscle function. Am J of Physiol, Cell Physiology 298: C149-C162
17. Tsvilovskyy, V.V., Zholos, A.V., Aberle, T., Dietrich, A., Zhu, M.X., Birnbaumer, L., Freichel, M., Flockerzi, V. (2009). Deletion of TRPC4 and TRPC6 in mice impairs smooth muscle contraction and intestinal motility in vivo. Gastroenterology, 137: 1415-1424.
18. Garrison, S.R., Dietrich, A., Stucky, C.L. (2012). TRPC1 contributes to light-touch sensation and mechanical responses in low-threshold cutaneous sensory neurons. J Neurophysiol 107: 913-922.
19. Hartmann, J., Dragicevic, E., Adelsberger, H., Henning, H., Sumser, M., Abramowitz, J., Blum, R., Dietrich, A., Freichel, M., Flockerzi, V., Birnbaumer, L., Konnerth, A. (2008). TRPC3 channels are required for synaptic transmission and motor coordination. Neuron 59, 392-398.
20. Beis, D., Schwarting, R.K., Dietrich, A. (2011). Evidence for a supportive role of classical transient receptor potential 6 (TRPC6) in the exploration behaviour of mice. Physiol Behav, 102: 245-250.
21. Griesi-Oliveira, K., Acab, A., Gupta, A.R., Sunaga, DY., Chailangkarn, T., Nicol, X., Nunez, Y., Walker, M.F., Murdoch, J.D., Sanders, S.J., Fernandez, T.V., Ji, W., Lifton, R.P., Vadasz, E., Dietrich, A., Pradhan, D., Song, H., Ming, G-l, Guoe, X., Haddad, G., Marchetto, M.C.N., Spitzer, N., Passos-Bueno, M.R., State, M.W., Muotri, A.R. (2015). Modeling non-syndromic autism and the impact of TRPC6 disruption in human neurons. Mol Psychiatry 20: 1350- 1365
All References
at PubMed

Fig. 5: The Dietrich Lab in the year 2020 from left to right: Bettina Braun, Alexander Dietrich, Fabienne Geiger, Suhasini Rajan, Christian Schremmer und Philipp Alt.

